Product Liability: Don’t be fooled by advertising, the nipple aspirate test doesn’t screen for cancer, the mammogram does!
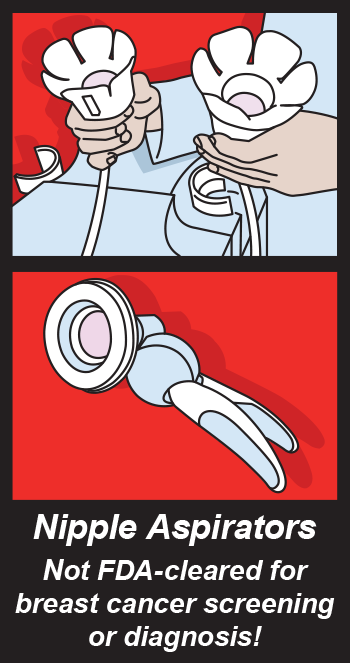 Some companies recently advertised the Nipple Aspirate Test as a method for breast cancer screening. The Nipple AspirateTest consists of using a breast pump to collect fluid from a woman’s nipple to screen for breast cancer. According the the FDA there is absolutely no clinical evidence to support these claims. So far the mammogram, even though it is uncomfortable for women, is the only way to screen for breast cancer.
Some companies recently advertised the Nipple Aspirate Test as a method for breast cancer screening. The Nipple AspirateTest consists of using a breast pump to collect fluid from a woman’s nipple to screen for breast cancer. According the the FDA there is absolutely no clinical evidence to support these claims. So far the mammogram, even though it is uncomfortable for women, is the only way to screen for breast cancer.
The company that is promoting this false advertising is Atossa Genetics. Atossa Genetics was issued a warning by the FDA that their test test was misbranded in that its labeling was false or misleading and asking them to address the violation. Atossa Genetics voluntary recalled the ForeCYTE Breast Health Test from the market in October 2013.
In its advertising Atossa Genetics falsely claimed that the test was “literally a Pap smear for breast cancer.”
The FDA recommends that all women who only did a nipple aspirate test for breast cancer should get a mammogram as soon as possible
 New York Personal Injury Attorneys Blog
New York Personal Injury Attorneys Blog


