Published on:
A company selling products intended for medical training purposes is recalling some of its practice IV Bags as reports came out that they were being used on patients
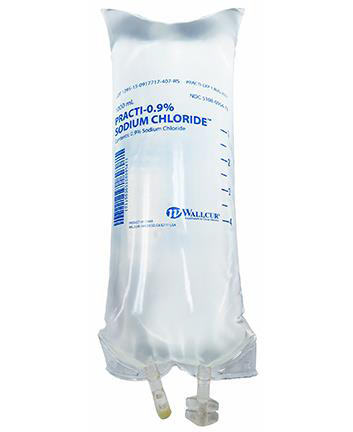
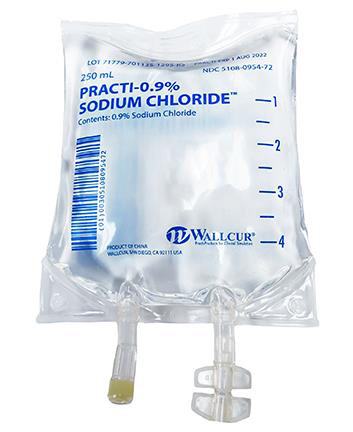
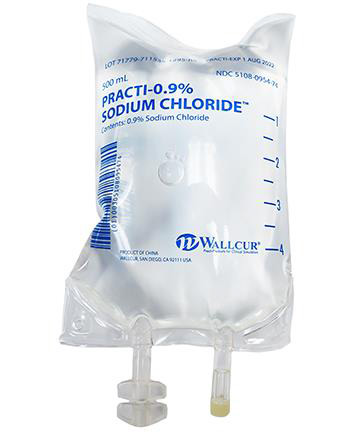
To administer non sterile products intended for training to patients is medical malpractice that can have very dangerous consequences. Recently Wallcur, a company that sells medical products intended for training purposes only, received reports that some of its training Sodium Chloride IV Bags had been used outside of their intended use and administered to patients. These bags are not sterile and are for simulation purposes only. To prevent these products from being injected in humans or animals, the company recently issued a product recall and is intending to improve the labels before to continue the distribution of these products..
 New York Personal Injury Attorneys Blog
New York Personal Injury Attorneys Blog


