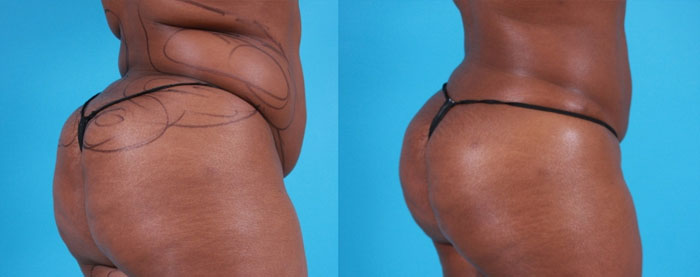
The death of a woman caused by silicon injections in her buttock was ruled a homicide by the New York City Medical Examiner
34 year old Kelly Mayhew died from medical malpractice last May in New York. She had traveled from Maryland with her mom to get cosmetic silicone injections of her buttock in Queens New York, City. Unfortunately, the doctor who injected her was an unlicensed phony plastic surgeon who fled the scene soon after Mayhew started to gurgle and struggle to breathe leaving her victim to die. The fake doctor never surrendered and the NYPD believe she know lives in England. The case is currently being presented to a grand jury in front of which the city Medical Examiner said that the cause of the death was “systemic silicone emboli due to cosmetic silicone injections of buttocks” and “the manner of death was homicide”. Read more in the NY Daily News
 New York Personal Injury Attorneys Blog
New York Personal Injury Attorneys Blog