Baxter Novum IQ Pump Recall: What Parents Should Know if Their Infant Was Exposed to This Dangerous Device
 Infants in neonatal intensive care units rely on precise intravenous infusions for survival—but a recent recall by Baxter International reveals that a widely used infusion pump may be putting vulnerable newborns at risk.
Infants in neonatal intensive care units rely on precise intravenous infusions for survival—but a recent recall by Baxter International reveals that a widely used infusion pump may be putting vulnerable newborns at risk.
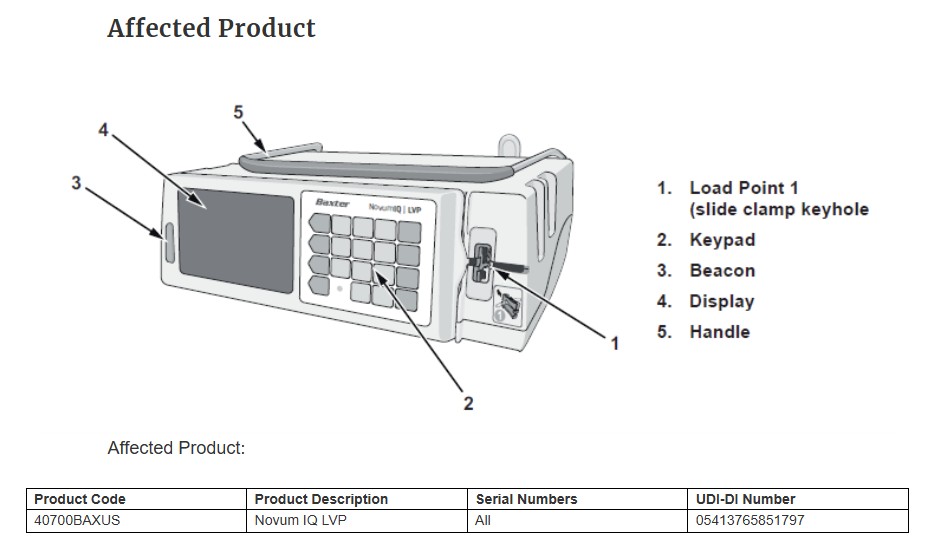
Baxter has issued a Class I recall—the FDA’s most serious designation—for its Novum IQ large volume pump after the device was linked to a serious injury caused by under-infusion. According to the FDA, variability of just 10% in infusion delivery can lead to dehydration, malnutrition, inadequate drug therapy, and even death in infants. Yet Baxter has allowed over 34,500 affected units to remain in hospitals across the U.S. and Canada.
 New York Personal Injury Attorneys Blog
New York Personal Injury Attorneys Blog







 Our firm is proud to announce that for the 13th consecutive year it has been named by U.S. News – Best Lawyers ® “Best Law Firms” a Tier 1 Firm in New York City for:
Our firm is proud to announce that for the 13th consecutive year it has been named by U.S. News – Best Lawyers ® “Best Law Firms” a Tier 1 Firm in New York City for:


